
Hi. My name is Gita and I have been a Therapeutic Radiographer for nearly 30 years. I’d like to share my professional and personal experiences of treating both primary and secondary breast cancer.

Hi. My name is Gita and I have been a Therapeutic Radiographer for nearly 30 years. I’d like to share my professional and personal experiences of treating both primary and secondary breast cancer.
METUPUK welcomes the approval of Piqray (alpelisib) plus fulvestrant for the treatment of hormone receptor-positive, HER2-negative, PIK3CA- mutated advanced breast cancer.
Piqray is an exciting new cancer medicine because it is the first targeted treatment option for advanced breast cancer that has a PIK3CA mutation.
METUPUK are delighted that NICE has accepted Trodelvy (sacituzumab govitecan) for routine use on the NHS in England for the treatment of patients with unresectable locally advanced or metastatic triple-negative breast cancer who have received two or more prior lines of systemic therapies. This follows on from the decision to approve Trodelvy in NHS Scotland in March 2022. Wales and Northern Ireland normally follow decisions by NHS England and we hope that Trodelvy will be made available to every patient who is eligible without delay.

Have you ever wondered what the relationship is like between primary and secondary breast cancer patients when they are talking about cancer?
We’ve put 4 of our MetUpUK members on a zoom call to chat it out – Mary and Laura have secondary cancer and Helen and Emily have had primary breast cancer.
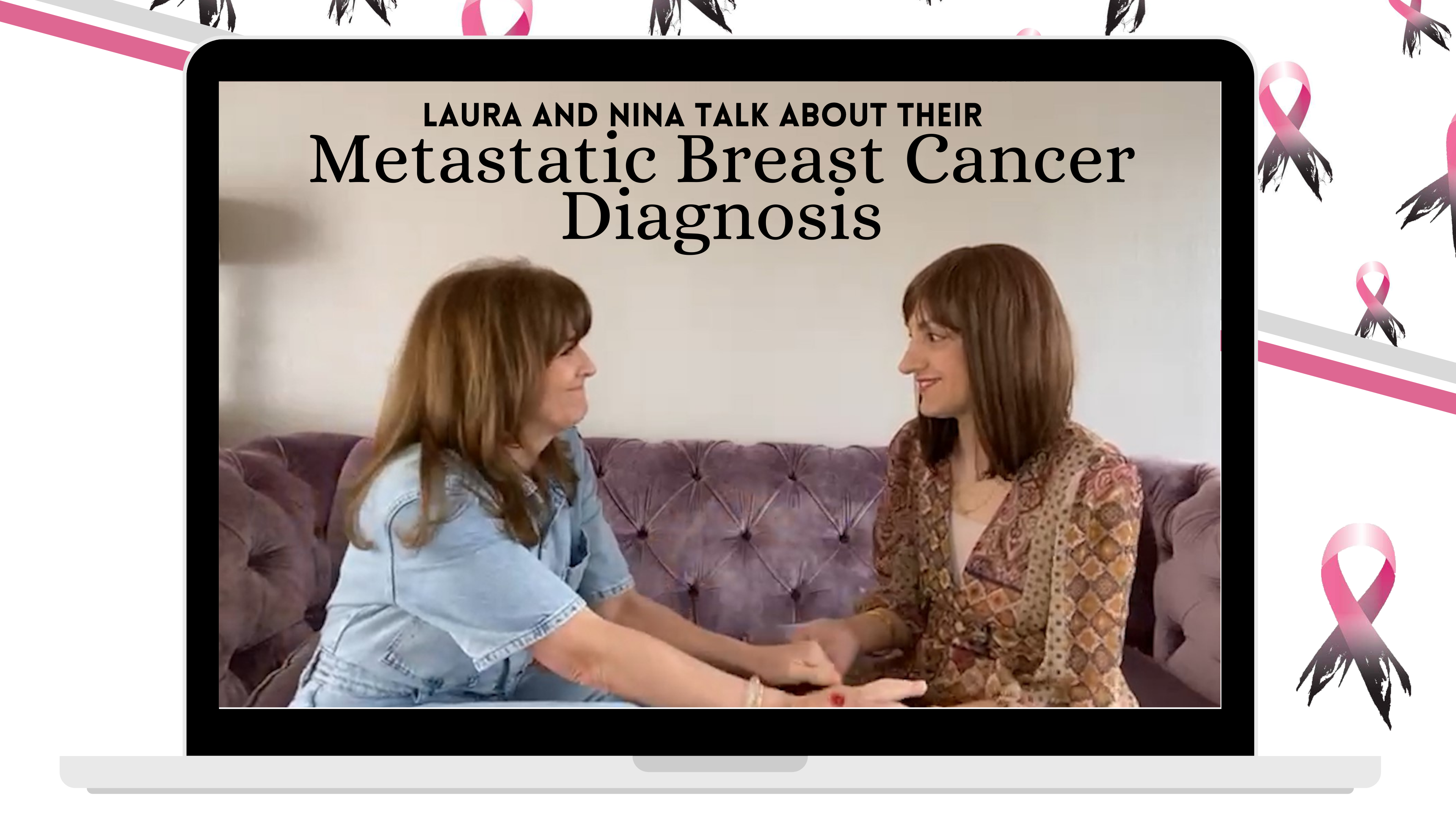
Laura has been living with metastatic breast cancer for 14.5 years, and Nina was diagnosed less than a year ago.
They have been friends since they were teenagers.
Join them as they chat about finding out their cancer was incurable.

31 life size figures of women are now set to tour the UK as an interactive campaign to raise awareness of issues surrounding secondary breast cancer after a small residency with the support of The Corn Exchange in Manchester.
When I first started volunteering with METUPUK, I wasn’t really sure what I would do. For a few months, I sat on the sides to get a feel for the organisation. I shared METUPUK links on social media, but I don’t have much of a social media presence, and Instagram is a bit of a mystery to me. I’ve never figured out how to do makeup, plus my hair is a non-event after chemo. I thought I wanted to help with drug access or access to clinical trials, but I was flexible and wanted to see what was needed.
To follow on from yesterday’s post about the death of Leila, I would like to highlight some information about brain metastases and just a quick look on google provided this information.
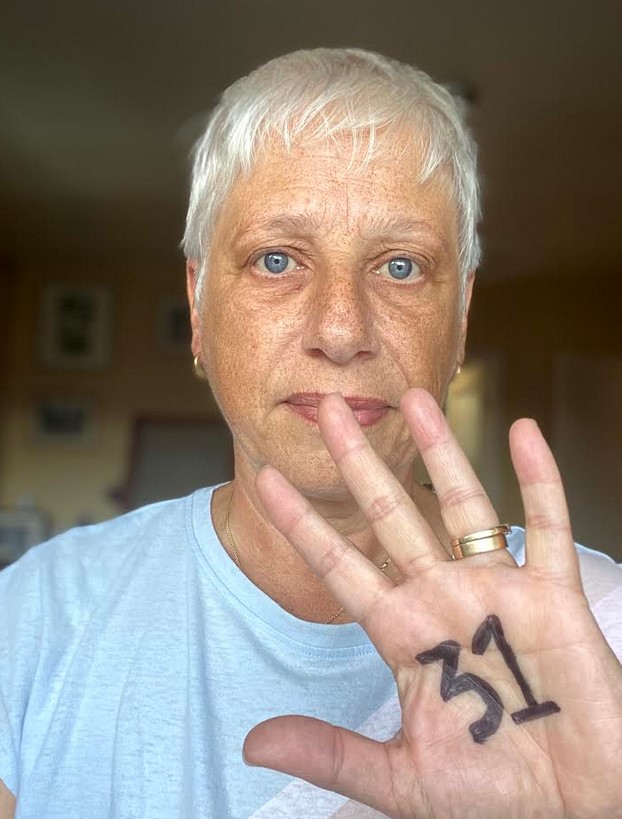
I was diagnosed with Grade 3 primary breast cancer in my left breast in January 2016 and am now cancer free. That sounds great to me. I’m one of the
It was shocking to hear the Prime Minister dismiss the importance of cancer outcomes in such an offhand way in a TV interview this week. But at the same time, it was sadly not surprising to many of us at Met-Up UK.