
I watched with interest the phenomenal feat by Lucy Gossage founder of 5K Your Way (Move Against Cancer podcast) and winner of the 2025 Spine Race which is an ultra

I watched with interest the phenomenal feat by Lucy Gossage founder of 5K Your Way (Move Against Cancer podcast) and winner of the 2025 Spine Race which is an ultra

We are very much minding the gap! Breast Cancer is the most diagnosed of ALL cancers worldwide. In the United Kingdom, Metastatic Breast Cancer is the biggest killer of working

Clinical trials are one of those things that no one really cares about until you need to. In the UK we have a pretty impressive cancer trial landscape that’s recognised

METUPUK’ Breast Cancer Awareness Month campaign for October across the UK and on social media channels. Continuing the synergy with The Darker Side of Pink travelling campaign highlighting the 31 women a

A cancer drug which received standing ovation from the Global Medical Community is being withheld from NHS breast cancer patients. METUPUK are devastated by NICE decision not to recommend the drug
METUPUK has joined the Northern Ireland Coalition (NICCC) and will work collaboratively with partner charities to improve outcomes for cancer patients in Northern Ireland. Ann McBrien, METUPUK’s Northern Ireland Executive
Just shy of six years ago I was diagnosed with Inflammatory Breast Cancer (IBC). Rare with a “high fatality rate and difficult to treat” is what I learnt in the
Here at METUPUK, we know drug access is a big issue for all metastatic breast cancer patients. There have been huge leaps and bounds made in terms of research, knowledge

METUPUK (Metastatic Exchange To Unleash Power) attended the 2024 United Kingdom’s Interdisciplinary Breast Cancer Symposium in Birmingham, hosted by Breast Cancer Now and supported by numerous partners. It was a
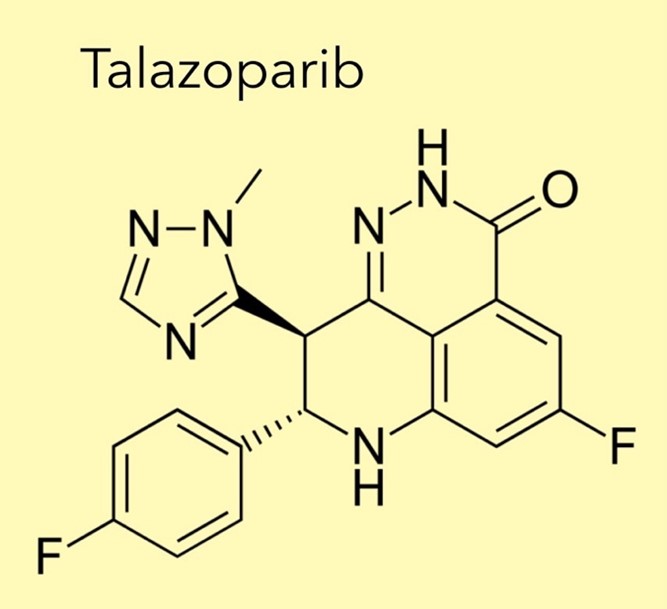
METUPUK are disappointed by the National Institute of Clinical Excellence (NICE) decision to not recommend talazoparib (Talzenna ®, Pfizer Ltd.) for treating HER2-negative, locally advanced or metastatic breast cancer with